Beyond Neurodegeneration: Functional Signatures of Parkinson’s Disease with HD-MEAs
Understanding Parkinson’s Disease Through Neuronal Function
Parkinson’s disease (PD) is a progressive neurodegenerative disorder affecting ~10 million people worldwide. It is classically defined by the selective loss of dopaminergic neurons in the substantia nigra pars compacta, leading to dopamine depletion in the striatum (Kalia & Lang, 2015).1 At the molecular level, PD is characterized by the misfolding and aggregation of α-synuclein into Lewy bodies and Lewy neurites (Surmeier et al., 2017)2.
Clinically, Parkinson’s disease manifests as:
- Motor symptoms: tremor, rigidity, bradykinesia, driven by striatal dopamine depletion
- Non-motor symptoms: cognitive impairment, mood disorders and autonomic dysfunction
- Circuit-level dysfunction: impaired basal ganglia activity regulating movement
However, beyond these defining features, a consistent pattern emerges: PD is driven as much by neuronal dysfunction as by degeneration. At a systems level, it is fundamentally a disorder of neuronal activity and circuit dynamics. Key functional features include:
- Altered firing patterns in dopaminergic circuits including loss of tonic dopaminergic neuron firing and disrupted regularity of firing
- Disrupted network synchronization and oscillations including network level irregular synchronicity after dopaminergic loss or altered burst dynamics
- Progressive impairment of neuronal connectivity and axonal signaling
These functional changes drive circuit-level dysfunction and ultimately clinical symptoms, making functional readouts essential for understanding disease mechanisms and detecting early-stage dysfunction.
From Clinical Phenotypes
to Functional Measurement
To connect clinical symptoms and circuit-level deficits to human-relevant models, in vitro systems must capture not only molecular pathology, but also neuronal function. The intrinsic cellular diversity, complex connectivity, and limited regenerative capacity of the human brain further complicate therapeutic development compared to other organ systems. Human iPSC-derived dopaminergic neurons as well as organoids and assembloids modeling nigrostriatal circuity, provide powerful platforms to recapitulate key aspects of PD. These systems reproduce altered neuronal activity, disrupted network coordination, and impaired connectivity, features that can emerge prior to overt neurodegeneration or neuronal loss (Stern et al., 2022).3
However, these functional phenotypes are not fully captured by conventional molecular or imaging assays. This creates a critical translational gap: drug candidates are often selected and advanced based on molecular or viability-based endpoints, while clinical efficacy in patients is ultimately determined by functional improvement. As a result, compounds that show promising molecular effects may fail to restore neuronal function at the circuit level, contributing to high attrition rates in PD drug development.
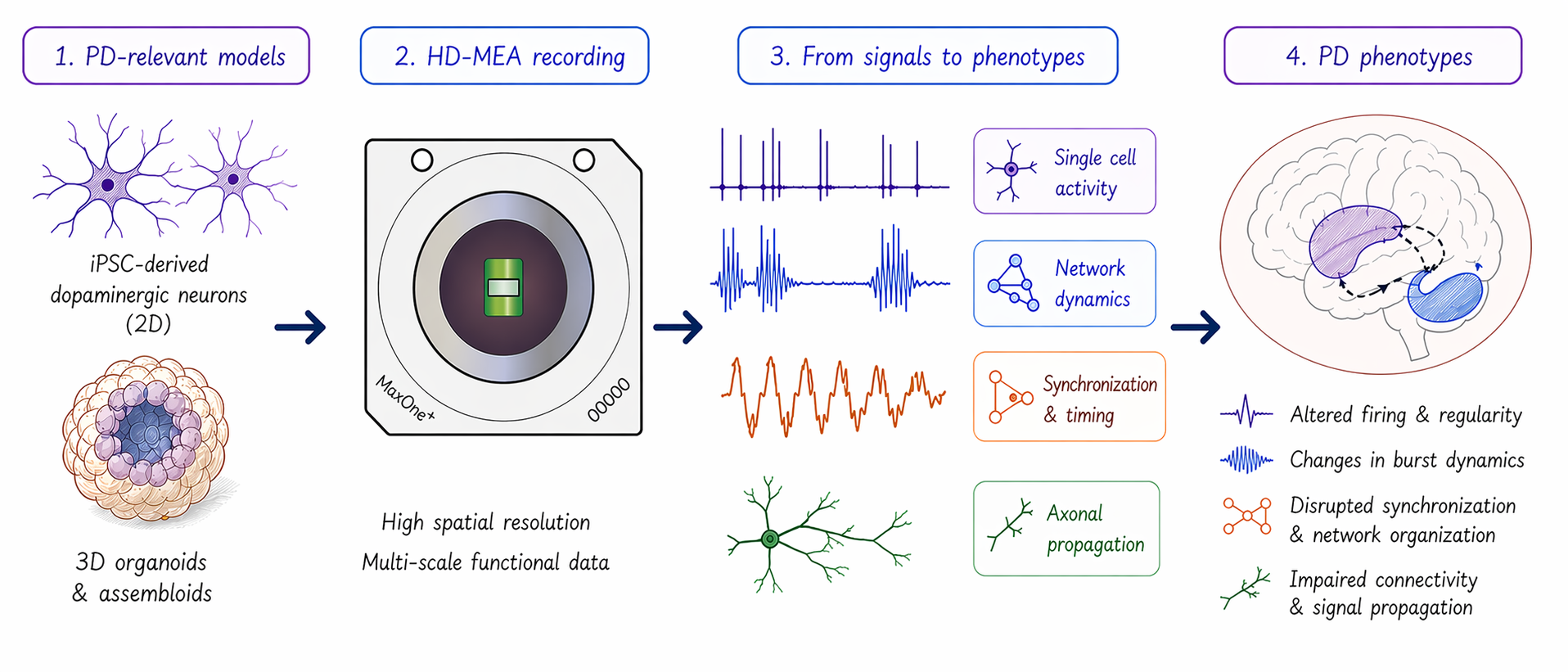
Closing this gap requires direct measurement of neuronal activity and network dynamics. High-density microelectrode arrays (HD-MEAs) address this need by enabling label-free functional readouts in human-relevant models to capture:
- Neuronal excitability: how neurons fire
- Temporal structure of activity: how firing patterns evolve
- Network coordination: how neurons synchronize
- Signal propagation: how activity travels across circuits
These HD-MEA measurements directly reflect functional deficits observed in PD patients and provide a critical link between in vitro models and clinical outcomes (Figure 1).
Functional Phenotypes of
Parkinson’s Disease Captured
with HD-MEAs
Across PD models, functional alterations extend beyond simple increases or decreases in activity. Instead, they reflect changes in timing, regularity, and network organization. MaxWell Biosystems’ HD-MEA platforms uniquely enable comprehensive neuronal characterization of these features across multiple biological scales by combining high spatiotemporal resolution with the ability to record activity from thousands of neurons simultaneously. This is enabled through three complementary assays:
- Network Assay: captures network-level dynamics, including synchronization, burst structure, and temporal organization
- ActivityScan Assay: provides whole sample activity maps of firing rate, spike amplitude, and firing regularity
- AxonTracking Assay: enables direct measurement of axonal signal propagation and connectivity
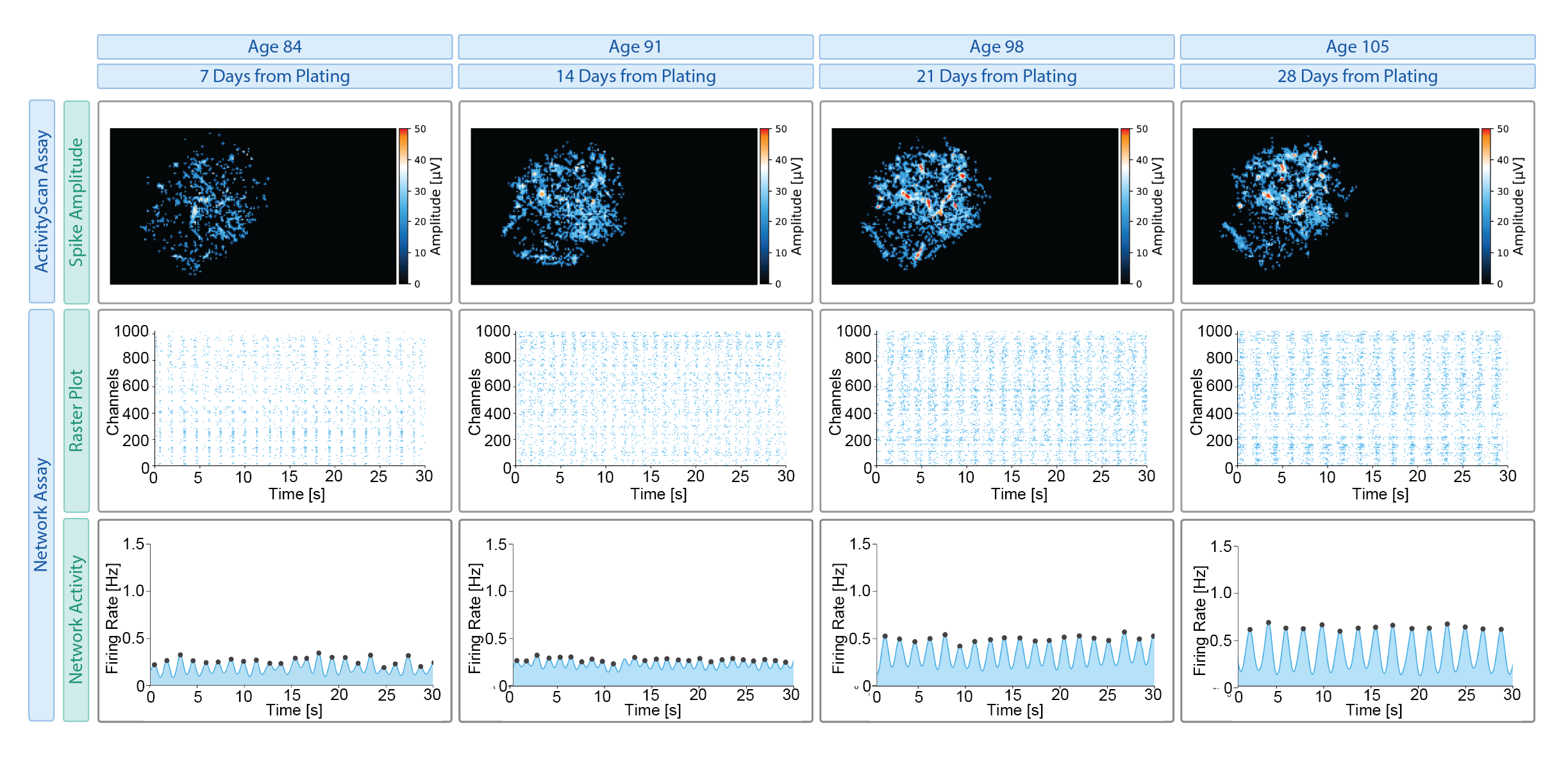
Together, these assays allow functional phenotyping from individual neurons to distributed networks and axonal pathways, capabilities that are difficult to achieve simultaneously with conventional electrophysiology or imaging approaches. These electrophysiological phenotypes can be characterized across diverse 2D and 3D PD-relevant models, including α-synuclein mutations (e.g., A53T, SNCA triplication), genetic risk variants (e.g., GBA, LRRK2) and perturbation-based models (Figure 2).
What are the key functional metrics
to quantify PD in vitro?
The table below provides a practical framework of robust, literature-supported functional readouts that can be directly extracted using MaxWell Biosystem’s HD-MEA Platforms.
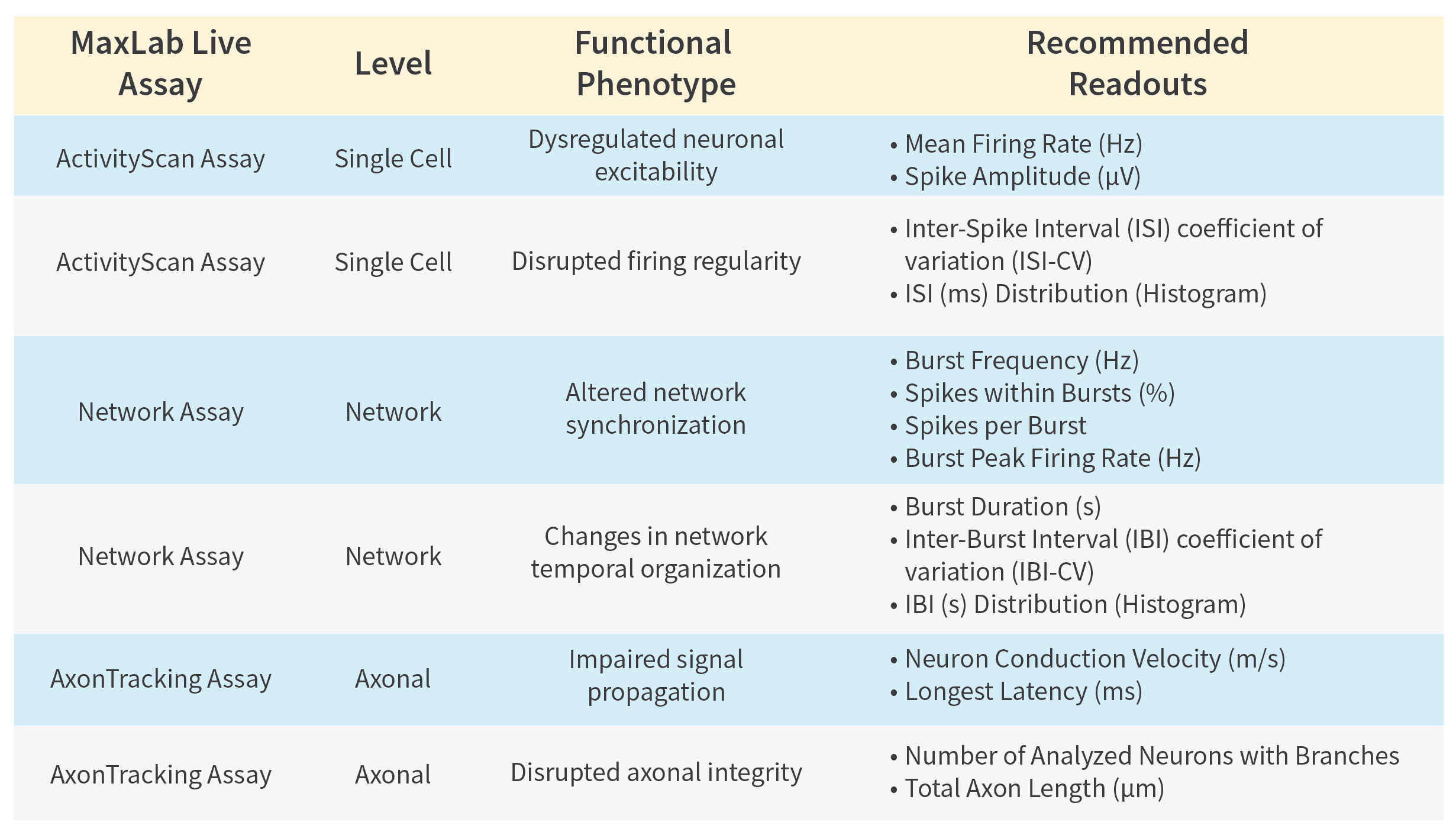
This multi-scale resolution enables:
- Detection of early, subtle functional deficits prior to degeneration
- Reproducible quantitative comparison between healthy and disease models
- High-throughput functional assessment of therapeutic interventions
Functional Evidence from HD-MEA
Studies in Parkinson’s Models
Recent publications using MaxWell Biosystems HD-MEA platforms demonstrate that the functional phenotypes outlined above are consistently observed across diverse PD-relevant models.

Tran et al., Brain, 2025
Developed human iPSC striatal-midbrain assembloids recapitulating α-synuclein propagation and nigrostriatal circuit dysfunction (SNCA overexpression). On the MaxOne platform, PD pathology led to decreased firing rate, reduced spikes per burst, and lower burst peak firing rate. These functional deficits correlated with loss of nigrostriatal projections, linking electrophysiology to circuit degeneration. Drug treatment restored both aggregation and functional activity, demonstrating utility for therapeutic screening.
Chaplot et al., bioRxiv, 2025
Patterned human iPSC-derived A9-like dopaminergic neurons and organoids exhibit tonic firing, increased firing rate, synchronized network activity, and measurable axonal conduction velocity on the MaxOne. Establishes a functionally mature, PD-relevant baseline model enriched for vulnerable SNpc-like subtypes.
Hornauer et al., Stem Cell Reports, 2024
In human iPSC-derived dopaminergic neurons (A53T vs control) on the MaxTwo 6-Well Plate, the PD model showed increased firing variability (↑ ISI-CV) and earlier, more frequent bursting. Network features such as inter-burst interval and periodicity provided strong separation between genotypes. This highlights network-level dysfunction as a key PD electrophysiological phenotype.
Hornauer et al., bioRxiv, 2022
Identified a reproducible electrophysiological fingerprint on the MaxOne platform in human iPSC-derived dopaminergic neurons with A53T mutation, including early emergence of network bursting and strong changes in network regularity frequency. Notably, α-synuclein downregulation induced a PD-like functional phenotype, linking α-syn biology to network dysfunction.
Ronchi et al., Advanced Biology, 2021
On the MaxOne platform, human iPSC-derived dopaminergic neurons with A53T SNCA mutation exhibited reduced firing rates, altered spike amplitude and more regular firing (↓ ISI-CV). Distinct network burst phenotypes included shorter network burst durations, shorter inter-burst intervals, and increased burst frequency. PD-related dysfunction reflects a reorganization of temporal firing structure, not just reduced neuronal activity.
Conclusion
PD is not only a disorder of neuronal loss, but of early and progressive dysfunction in neuronal activity and circuit dynamics. MaxWell Biosystem’s HD-MEA platforms provide a direct, label-free approach to measure these changes as they emerge. By capturing electrophysiological phenotypes across scales, from single-cell firing to network coordination and axonal signal propagation, HD-MEAs enable a deeper mechanistic and translational understanding of Parkinson’s disease.
For drug discovery, this distinction is critical. Therapeutic success in patients depends on restoring functional activity, not just modifying molecular pathology. MaxWell Biosystems’ HD-MEA platforms enable direct measurement of these functional phenotypes in human-relevant models, bridging the gap between preclinical assays and clinical endpoints. Integrating functional phenotyping into the drug discovery pipeline enables more predictive models, better-informed decision-making, and ultimately increases the likelihood of clinical success. h
Related
Resources
MaxTwo
Maximize your cell functional assays

MaxLab Live
All-in-one Software
Our Technology
Multi-/Micro-electrode Arrays

Disease Modeling

Functional Phenotyping
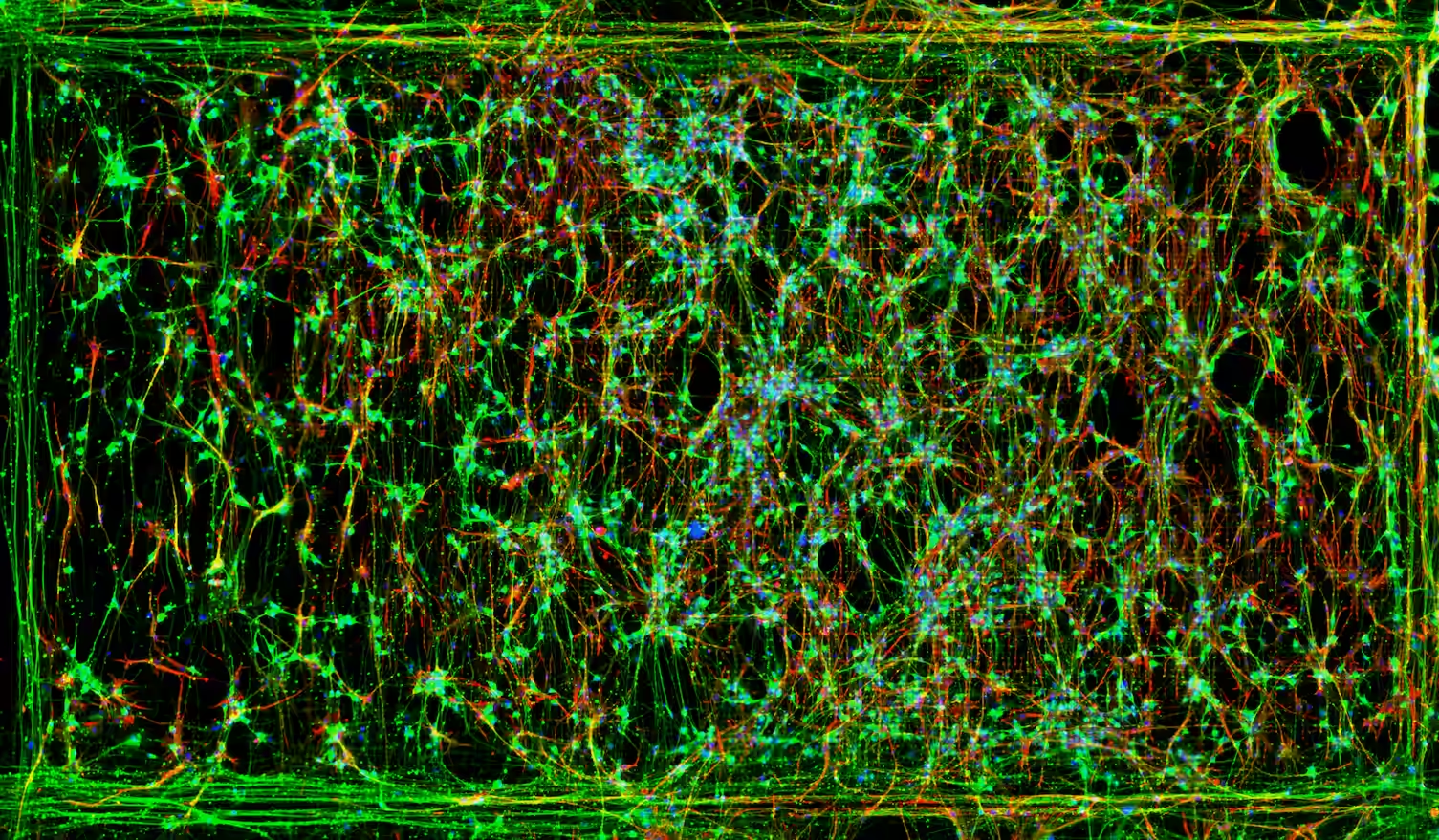
Neuronal Cell Cultures
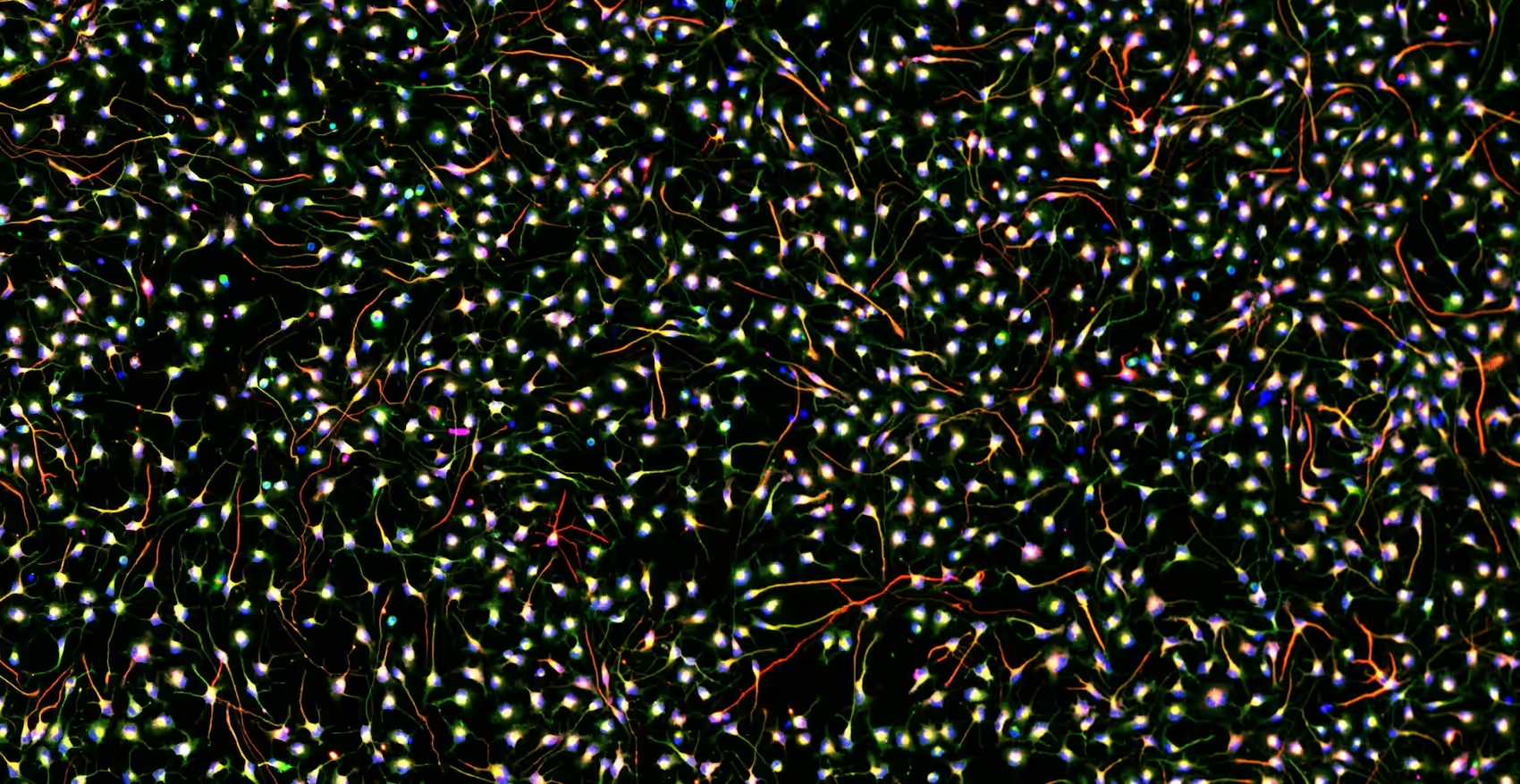
Organoids