ISSCR 2026
Date & Time
Location
Tags
ISSCR 2026 is one of the landmark annual meetings in stem cell research, bringing together the global community to share breakthroughs, exchange ideas, and explore the latest advances across the field. Covering topics from basic stem cell biology to disease modeling, drug discovery, engineering, and clinical translation, it is a key meeting for staying close to where stem cell research is heading.
As every year, we are excited to be there, connecting with the community, discussing new ideas, and support your research and next discovery. Join us at our booth, innovation showcase, and posters to discover how next-generation HD-MEA technology brings functional insights to stem cell research, and learn what's new in the field.

Meet the MxW Team
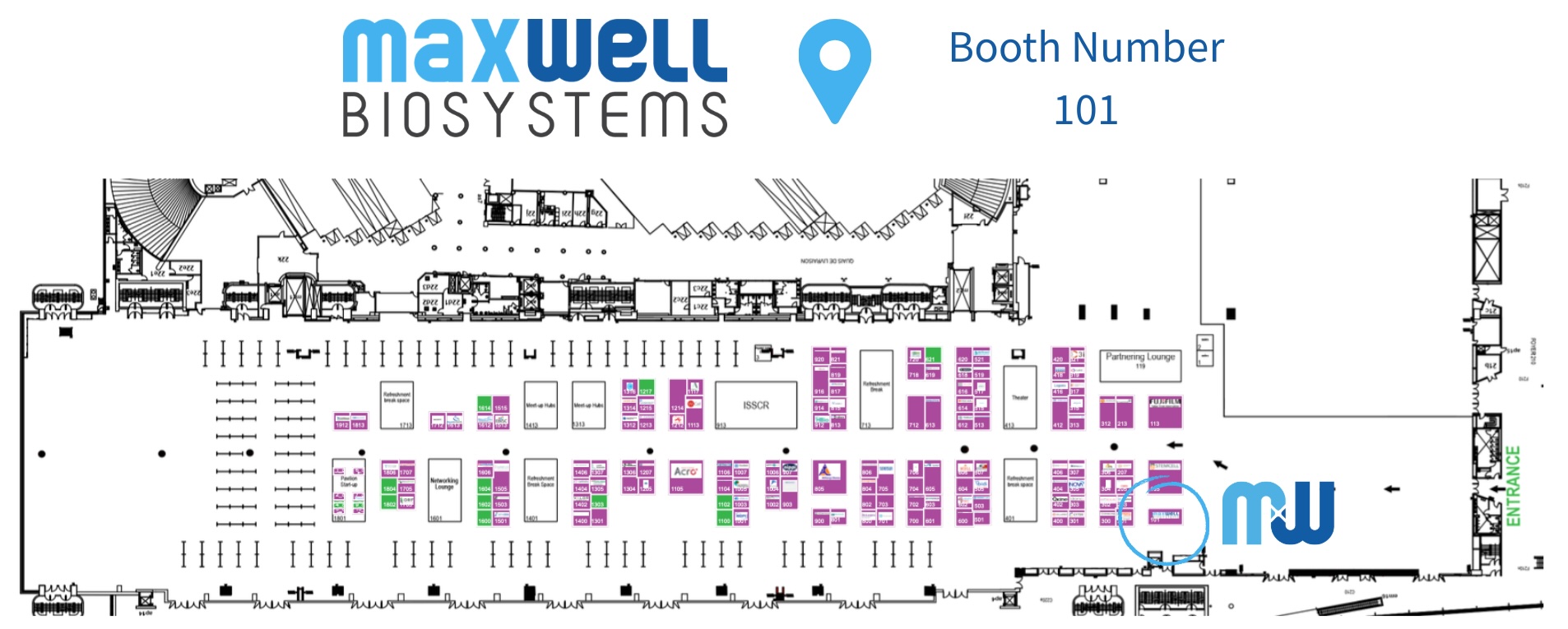
MxW Booth
Come visit us at booth 101 to find out how MaxWell Biosystems supports stem cell research with scalable high-content functional electrophysiology, delivering high-resolution insights and quality data you can trust. Stop by to connect with the team, explore what is new, and get a glimpse of what is coming next.
Innovation Showcase
Join us for this Innovation Showcase to explore scalable functional assays for disease modeling and drug discovery with iPSC-derived neural cultures and brain organoids, powered by next-generation HD-MEA technology. From recent platform advances to translational organoid research in space, the session will highlight new ways to capture functional insights from network to subcellular scale.
Register using the form below!
Agenda
Welcome & Introduction – Dr. Laura D’Ignazio
Recent Developments at MaxWell Biosystems – Dr. Marie Obien
Invited Talk – Dr. Aline Martins
Q&A Session – All speakers
Closing Remarks – Dr. Laura D’Ignazio
Poster Presentations
Workshops
Join us for this Innovation Showcase to explore scalable functional assays for disease modeling and drug discovery with iPSC-derived neural cultures and brain organoids, powered by next-generation HD-MEA technology. From recent platform advances to translational organoid research in space, the session will highlight new ways to capture functional insights from network to subcellular scale.
Register using the form below!
Agenda
Welcome & Introduction – Dr. Laura D’Ignazio
Recent Developments at MaxWell Biosystems – Dr. Marie Obien
Invited Talk – Dr. Aline Martins
Q&A Session – All speakers
Closing Remarks – Dr. Laura D’Ignazio




.png)

